Analysis of layer growth kinetics
Introduction
During real-time RBS one is measuring the thickness of some layer, ![]() , whose value is given by integrating the rate of change in
, whose value is given by integrating the rate of change in ![]() over suitable limits,
over suitable limits,
(1) ![]()
where the model of the growth of the layer is contained in the rate equation in square brackets. Eq. (1) is independent of the model, and can therefore be used for constant temperature as well as ramped temperature anneals, provided of course that an appropriate model is used. The initial conditions are expressed as ![]() and often it is taken that
and often it is taken that ![]() , but this is not necessary as long as the thickness at time
, but this is not necessary as long as the thickness at time ![]() is known.
is known.
The rate equation at constant temperature
Let ![]() denote the rate equation. When the temperature remains constant over time, one can model the growth with a very general model that incorporates a full description of the kinetics. However, it is not apparent which of the competing models should be used. Fortunately, one almost always finds the limiting behaviour of either diffusion controlled growth or in some rare cases linear growth for film thicknesses in the range where RBS analysis is suitable (
denote the rate equation. When the temperature remains constant over time, one can model the growth with a very general model that incorporates a full description of the kinetics. However, it is not apparent which of the competing models should be used. Fortunately, one almost always finds the limiting behaviour of either diffusion controlled growth or in some rare cases linear growth for film thicknesses in the range where RBS analysis is suitable (![]() nm).
nm).
Diffusion controlled kinetics
In the case of diffusion controlled kinetics, the growth rate is inversely proportional to the thickness of the growing layer and is given by
(2) ![]()
where
(3) ![]()
Linear reaction kinetics
In the case of linear reaction kinetics, the growth rate is assumed to be limited by the reactions taking place at the interface, and not on the rate at which they arrive (by diffusion, for example). Hence the rate equation is quite simply:
(4) ![]()
where
(5) ![]()
The rate equation in a linear ramp
During a typical experiment the temperature ramp rate, ![]() , is kept constant during the time interval
, is kept constant during the time interval ![]() , and
, and
(6) 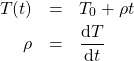
Since,
(7) ![]()
eq. (1) is transformed to
(8) ![]()
The rate constants, ![]() , used in diffusion controlled growth (2) and linear growth (4), at constant temperature, usually have a temperature activated dependence, and generally
, used in diffusion controlled growth (2) and linear growth (4), at constant temperature, usually have a temperature activated dependence, and generally
(9) ![]()
with ![]() an activation energy (in diffusion control it is the apparent activation energy for the interdiffusion, while in linear growth it is the activation energy of the corresponding chemical reaction),
an activation energy (in diffusion control it is the apparent activation energy for the interdiffusion, while in linear growth it is the activation energy of the corresponding chemical reaction), ![]() is Boltzmann’s constant and
is Boltzmann’s constant and ![]() is a constant that does not depend on time or temperature.
is a constant that does not depend on time or temperature.
The introduction of the temperature activated term into the rate equation in eq. (8) allows its solutions to be determined in terms of the exponential integral, ![]() .
.
typeset by QuickLaTeX